 
What does it matter pacheno valley? Do you have proof? Ok you are right! Now what? What changes? what are the consequences of your ideas? Everyone already knows that all physical models are idealized and therefore flawed. Best you communicate with people who really do care and really do have open minds. 
Entropy is both a dogma and a paradigm perpetuated by closed minds and the illustrious academia. Built a motor that recovers energy, best case just under 90%, no one believes that either, even seeing the thing run. Built a motor that removed reverse EMF almost completely, but no one picked up on that either. Won the bet, had full instrumentation on the gen, but still the other engineers ignored it. Built a generator using switched reluctance, property was no load to full load with no change in input power. Perhaps no understanding.ĭone those things myself. 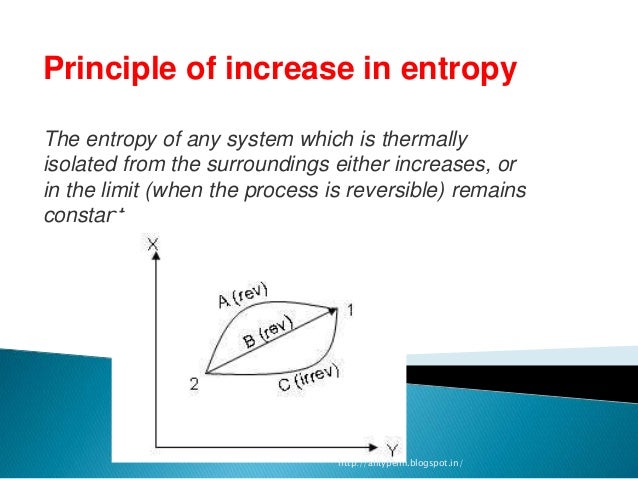
Started a thread here that gives methods used by others. These caused a small stir when printed, but died out through apparent lack of understanding.įor years, I have maintained that through magnetic functions we can utilize ZPE and reduce or eliminate fueled homes, vehicles, etc. Defeat of a current paradigm is near impossible.įor years, I have stated that there are ways to utilize the natural power of the universe, and wrote many papers on the orthorotation of ZPE through magnetic means. While I admire your efforts, you are whipping a dead horse.ĭefeat of dogma is terribly difficult. Pencho, I've said this before, but once again. for more than a century and the truth has become self-evident. Thermodynamicists know that and have adopted a specific tactic: they have repeated "Entropy is a state function", "Entropy is a state function" etc. The proof offered by Clausius is based on the assumption "A general cycle can be divided into small Carnot cycles" which is almost obviously wrong. Yet the premise "Entropy is a state function" has no serious justification. The importance of the second premise is even greater - just try to imagine modern science where all statements involving the concept of entropy are absurd. "Shatter this postulate, and modern physics becomes an elaborate farce!" The importance of the first premise is correctly expressed by late Bryan Wallace: The speed of light is constant, independent of the relative speed of the light source and the observer. Therefore, 3 mol of gas give 2 mol of liquid which means that there is a decrease in entropy.Roughly speaking, modern science is based on two premises:ġ. Here, m and n are the appropriate coefficients in the balanced chemical equation.Ĭonsider 2 H 2(g) + O 2(g) -> 2 H 2O(liq) Hence, Entropy change = Entropy Transfer + Entropy Generation Since the temperature T is constant, the two quantities of entropy along A and B+C are equal showing that change in entropy is equal while heat is not. In an isolated system,dQ = 0,which means that if the process occurs reversibly dS = 0 and irreversibly dS> 0.įigure : P-V graph of an expansion systemįrom the P-V graph above A: isothermal expansion, B:adiabatic expansion, C:isochoric process Thus, the more high the entropy generation the higher the irreparability and, accordingly, the lowest the efficiency of a device since a reversible system is the most efficient system.Įntropy is a state function which is independent of the path or history of the system. The generation of entropy is because of the presence of irreversibility. This means that the universe is always becoming disorganized and is approaching a chaotic state. Table 1: Predicting spontaneity and non-spontaneity of a systemĮntropy, unlike energy, is non-conservative because it is always increasing or remains constant but never decreases. Therefore,Īt equilibrium, ∆ S univ= ∆ S sys + ∆ S surr= 0 When a system reaches equilibrium, both forwardand the reverse reaction are non- spontaneous because there is no driving force. The second law of thermodynamics states that for an isolated or a closed adiabaticor System + Surroundings. 
Remember that –∆H˚ sys is proportional to ∆S˚ surr and an exothermic process has ∆S˚ surr> 0. In conclusion, S (gases) > S (liquids) > S (solids) In most cases, entropy becomes greater in size when a true solid or liquid close down in a solvent. Accordingly, an increase in molecular complexity results in an increase in S.Entropies of ionic solids depends on coulombic attractions. This means that more disorganized a system is, the higher its entropy.The entropy of a substance increases with temperature. Entropy is a thermodynamic property which is a measure of the disorder of a system. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
